Section 1: Electrons & Chemical Bonding
Vocabulary
1) Chemical Bonding: the combining of atoms to form molecules of ionic compounds
2) Chemical Bond: an interaction that holds atoms or ions together
3) Valence Electron: an electron that is found in the outermost shell of an atom and that determines the atom's chemical properties
2) Chemical Bond: an interaction that holds atoms or ions together
3) Valence Electron: an electron that is found in the outermost shell of an atom and that determines the atom's chemical properties
Section Summary
This section discussed electrons and chemical bonding. They just scratched the surface my describing what a chemical bond is and and what the result of a chemical bond will be (a new substance). Next, they discussed the basic structure of an atom and the atomic number (the number of protons within the nucleus). While discussing the basic structure of an atom they also spoke about the arrangement of electrons within an atom. The first level can hold up to two electrons, the second level can hold up to eight, and the third level can hold up to eight. The third or outer level/shell is unique because these electrons are the electrons that are lost or gained when a chemical bond takes place. Atoms will take and give away valence electrons to have an even number of electrons in its outer levels.
Section 2: Ionic bonds
Vocabulary
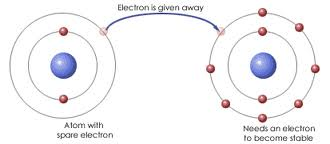
1) Ionic Bond: a bond that forms when electrons are transferred from one atom to another, which results in a positive ion and a negative ion.
2) Ion: a charged particle that forms when an atom or group of atoms gains or loses one or more electrons.
3) Crystal Lattice: the regular pattern in which a crystal is arranged
2) Ion: a charged particle that forms when an atom or group of atoms gains or loses one or more electrons.
3) Crystal Lattice: the regular pattern in which a crystal is arranged
Section Summary
In this section they discussed ionic bonds. During an ionic bond (vocabulary word 1),one or more valence electrons are transferred from one atom to another. Next, they discussed charged particles. When an atom gains electrons then it becomes negatively charged, but when it loses electrons it becomes positively charged (but a large amount of energy is needed to pull electrons from atoms). Also, the number of electrons gained or lost depends on the number of electrons in the outer level/shell. Nonmetal atoms gain electrons very easily due to the fact that the outer level is almost full. The more easily an atom gains an electron the more energy it releases. Lastly, when ions (vocabulary word 2) bond, they form a repeating three-dimensional pattern called a crystal lattice (vocabulary word 3).
Helpful Site Just a Click Away! Ionic Bonding
Section 3: Covalent & mettalic bonds
Vocabulary
1) Covalent Bond: a bond formed when atoms share one or more pairs of electrons
2) Molecule: the smallest unit of a substance that keeps all of the physical and chemical properties of that substance
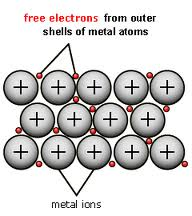
3) Metallic Bond: a bond formed by the attraction between positively charged metal ionsand the electrons around them
2) Molecule: the smallest unit of a substance that keeps all of the physical and chemical properties of that substance
3) Metallic Bond: a bond formed by the attraction between positively charged metal ionsand the electrons around them
Section Summary
This section discussed covalent and metallic bonds. Covalent bonds (Vocabulary word one) only occur between nonmetals due to the fact that transferring electrons would require too much energy. Covalently bonded nonmetal atoms form molecules (Vocabulary word two), for example, a water molecule (the smallest particle that has the chemical properties of water) is composed of two hydrogen atoms and one oxygen atom. Also, substances that are covalently bonded usually have a low melting and boiling point and are brittle in their solid state.
Next, are simple and complex molecules. Simple molecules include diatomic molecules and diatomic elements. Diatomic molecules are molecules made up of two atoms. Diatomic elements are elements that are found in nature as diatomic molecules. More-complex molecules are made of three or more atoms. Carbon atoms are the basis of many of these complex molecules.
Lastly, are metallic bonds (Vocabulary word one). In a metallic bond electrons move freely and keep ions together. Metallic bonds extend throughout the metal in all directions. Metallically bonded substances have similar properties including good conductors of electric current, easily reshaped (malleability[flattened out] & ductility[strung into thin wire]), and able to be bent without breaking.
Next, are simple and complex molecules. Simple molecules include diatomic molecules and diatomic elements. Diatomic molecules are molecules made up of two atoms. Diatomic elements are elements that are found in nature as diatomic molecules. More-complex molecules are made of three or more atoms. Carbon atoms are the basis of many of these complex molecules.
Lastly, are metallic bonds (Vocabulary word one). In a metallic bond electrons move freely and keep ions together. Metallic bonds extend throughout the metal in all directions. Metallically bonded substances have similar properties including good conductors of electric current, easily reshaped (malleability[flattened out] & ductility[strung into thin wire]), and able to be bent without breaking.
Helpful Site Just a Click Away! Covalent & Metallic Bonding