Section 1: Radioactivity
Vocabulary
1) Radioactivity: the process by which an unstable nucleus gives off nuclear radiation
2) Mass Number: the sum of the numbers of protons and neutrons in the nucleus of an atom
3) Isotope: an atom that has the same number of protons (or the same atomic number) as other atoms of the same element do but that has a different number of neutrons (and thus a different atomic mass)
4) Half-Life: the time needed for half of a sample of a radioactive substance to undergo radioactive decay
2) Mass Number: the sum of the numbers of protons and neutrons in the nucleus of an atom
3) Isotope: an atom that has the same number of protons (or the same atomic number) as other atoms of the same element do but that has a different number of neutrons (and thus a different atomic mass)
4) Half-Life: the time needed for half of a sample of a radioactive substance to undergo radioactive decay
section summary
This section discussed radioactivity. Radioactivity (vocabulary word 1) was first discovered (according to modern history) by a French scientist named Henri Becquerel. Becquerel discovered radiation in an experiment where he placed a flourescent mineral behind a photographic plate and let sunlight shine through it. In doing this he could see energy being released from the mineral (radioactivity). Later one of Henri's co-workers named the process by which some nuclei give off nuclear radiation radioactive decay.
There are three kinds of radioactive decay. The first is alpha decay, which is the release of an alpha particle from a nucleus. It has a mass number (vocabulary word 2) of 4 and a charge of 2+. Alpha particles are the largest of all radioactive particles and do the least damage (can be stopped by paper or clothing). Next is beta decay, which is when a beta particle is released from the nucleus. It has a mass number of zero because it has no protons or neutrons. There are two types of beta decay carbon-14 and carbon-11. In carbon-14 a neutron breaks into a proton and an electron and mass number and charge are conserved. Also, it becomes an entirely different element. In carbon-11 a proton breaks into a positron and a neutron. At this point it is now an entirely different element. Both of these atoms are also isotopes (vocabulary word 3). Beta particles are the second strongest of all of the radioactive particles and can be stopped by aluminum.
Lastly, the strongest form of radioactive decay is gamma decay, which is when gamma rays are released from the nucleus. Gamma rays are very high levels of energy are given off during alpha or beta decay. Gamma rays have no mass and no charge. They are the smallest of radioactive particles but penetrate very deep and can only be stopped by very dense substances (led, concrete, etc.). Gamma decay does not cause a change in elements. A single large exposure to any one of these forms of radiation however, can lead to radiation sickness.
You can also find the age of different things by measuring the number of half-lives (vocabulary word 4) that have gone by. Different isotopes of elements have different half lives (Ex. Uranium-238: 4.5 billion years; Hydrogen-3: 12.3 years). After every half-life the amount of the substance is divided by two (1 half-life= 1/2, 2 half-lives=1/4, 3 half-lives= 1/8, etc.). Radioactivity also has other positives. In healthcares it is used to sterilize products and in industry it is used to detect defects in structures.
There are three kinds of radioactive decay. The first is alpha decay, which is the release of an alpha particle from a nucleus. It has a mass number (vocabulary word 2) of 4 and a charge of 2+. Alpha particles are the largest of all radioactive particles and do the least damage (can be stopped by paper or clothing). Next is beta decay, which is when a beta particle is released from the nucleus. It has a mass number of zero because it has no protons or neutrons. There are two types of beta decay carbon-14 and carbon-11. In carbon-14 a neutron breaks into a proton and an electron and mass number and charge are conserved. Also, it becomes an entirely different element. In carbon-11 a proton breaks into a positron and a neutron. At this point it is now an entirely different element. Both of these atoms are also isotopes (vocabulary word 3). Beta particles are the second strongest of all of the radioactive particles and can be stopped by aluminum.
Lastly, the strongest form of radioactive decay is gamma decay, which is when gamma rays are released from the nucleus. Gamma rays are very high levels of energy are given off during alpha or beta decay. Gamma rays have no mass and no charge. They are the smallest of radioactive particles but penetrate very deep and can only be stopped by very dense substances (led, concrete, etc.). Gamma decay does not cause a change in elements. A single large exposure to any one of these forms of radiation however, can lead to radiation sickness.
You can also find the age of different things by measuring the number of half-lives (vocabulary word 4) that have gone by. Different isotopes of elements have different half lives (Ex. Uranium-238: 4.5 billion years; Hydrogen-3: 12.3 years). After every half-life the amount of the substance is divided by two (1 half-life= 1/2, 2 half-lives=1/4, 3 half-lives= 1/8, etc.). Radioactivity also has other positives. In healthcares it is used to sterilize products and in industry it is used to detect defects in structures.
Helpful Site Just a Click Away! Radioactivity
section 2: Energy from the Nucleus
vocabulary
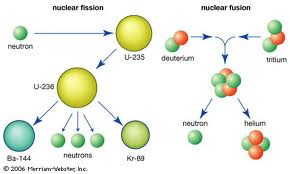
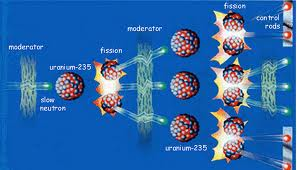
1) Nuclear Fission: the splitting of the nucleus of a large atom into two or more fragments; releases additional neutrons and energy
2) Nuclear Chain Reaction: a continuous series of nuclear fission reactions
3) Nuclear Fusion: the combination of the nuclei of small atoms to form a larger nucleus; releases energy
2) Nuclear Chain Reaction: a continuous series of nuclear fission reactions
3) Nuclear Fusion: the combination of the nuclei of small atoms to form a larger nucleus; releases energy
section summary
This section discussed energy from the nucleus. Energy can come from nuclear fusion (vocabulary word 3) and nuclear fission (vocabulary word 1). In nuclear fission a larger nuclei splits into two smaller nuclei. Sometimes when more and more neutrons fly apart and cause more nuclei to to explode, this is called a nuclear chain reaction (vocabulary word 2), however, if these reactions are not carefully controlled then they can lead to serious accidents (such as the Chernobyl, Ukraine accident). Nuclear fission also produces a lot of waste and cost more to build that fossil fuel power plants, but they do not release as many gases into the atmosphere. Nuclear fusion however is harder to process because it requires plasma (which is extremely hard to create because it requires more than 100,000,000 degrees Celsius). However, if one day humans are easily able to create nuclear fusion then it will revolutionize the way we do most things in our lives. Nuclear fusion is very efficient. It is less accident prone, there is more fuel availible (hydrogen-2 can be fused to create large amounts of energy), and the products of fusion are not radioactive so it's more eco-friendly.
Helpful Site Just a Click Away! Nuclear Fusion & Nuclear Fission